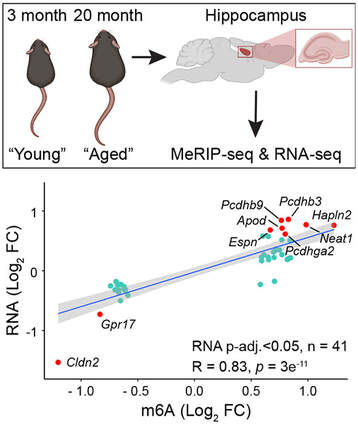
 Our new research describing the m6A-epitranscriptomic profiles from the hippocampi of young (3 month-old) vs aged (20 month-old) C57Bl/6 mice is now out in Aging Cell. We reveal more over 500 transcripts that are differentially methylated. There is also a significant concordance between m6A and transcript levels in both directions. Finally, we found that the myelin regulator gene Gpr17 was downregulated in the aged hippocampus concomitant with reduced m6A levels in its 3'UTR. Overall, the positive correlation between m6A and the transcript expression levels indicates a co-transcriptional regulation of m6A with gene expression changes that occur in the aged mouse hippocampus. Congratulations to River and Jocelyn who lead the project, as well as to our fantastic collaborators Drs. Renhua Song and Justin Wong (Centenary Institute, Sydney). We are delighted to that our new work is now published in Open Biology. Here we reports the structural basis of familial ALS-associated SFPQ variants in promoting the formation of SFPQ cytoplasmic aggregates through enhanced zinc binding. We also found that these aggregates alter the expression of AMPA receptors on the plasma membrane of primary neurons, a phenotype that is commonly found in ALS patients. This is another great collaboration with the lab of Mihwa Lee (La Trobe University) and is an extension of our previous work published in Nucleic Acids Research. Well done Jocelyn, Nishita, Anson and Lara for contributing to this study. Thanks to MND Research Australia (Judy Mitchell MND Research Innovator Grant) for supporting this research.
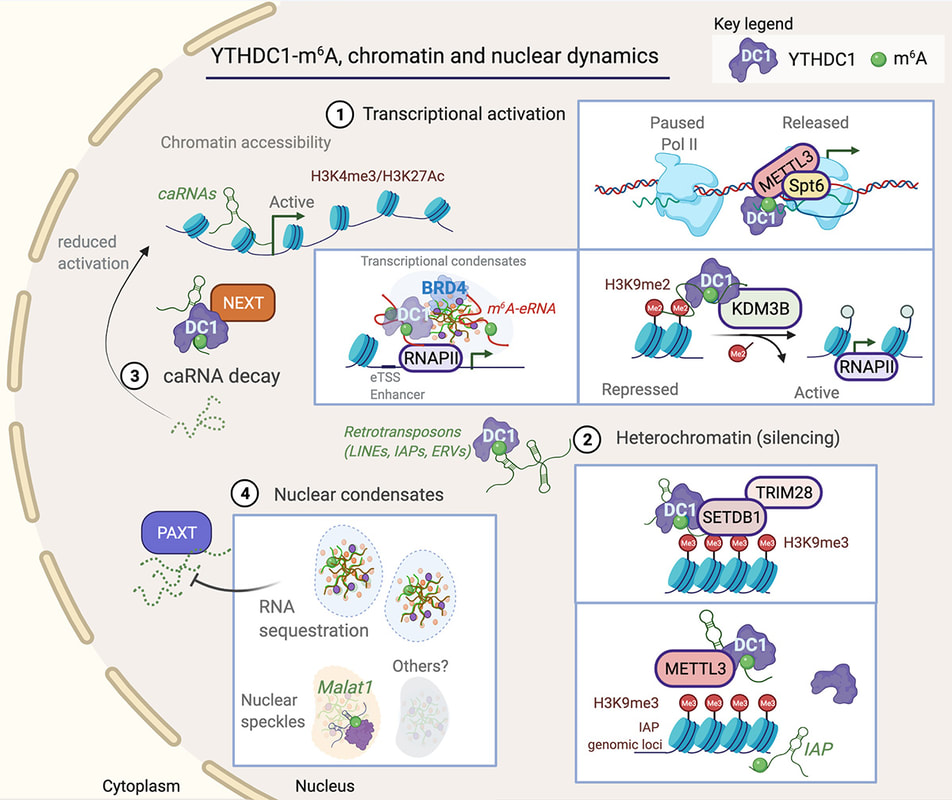
Jocelyn wrote an opinion piece that was published in Trends in Genetics. We propose a unifying mechanism that explains the complexity of YTHDC1 (the nuclear m6A reader) in regulating gene expression. Another great collaboration with Dr Justin Wong (Centenary Institute, Sydney).
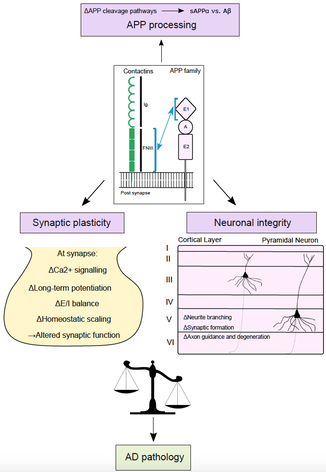
Happy to share our new review article by Jocelyn on the m6A regulatory mechanisms of brain plasticity, learning and memory, which has just been accepted for publication in Seminars in Cell and Developmental Biology. A great collaboration with Dr. Justin Wong (Centenary Institute, Sydney).
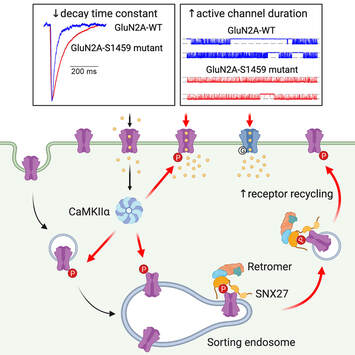
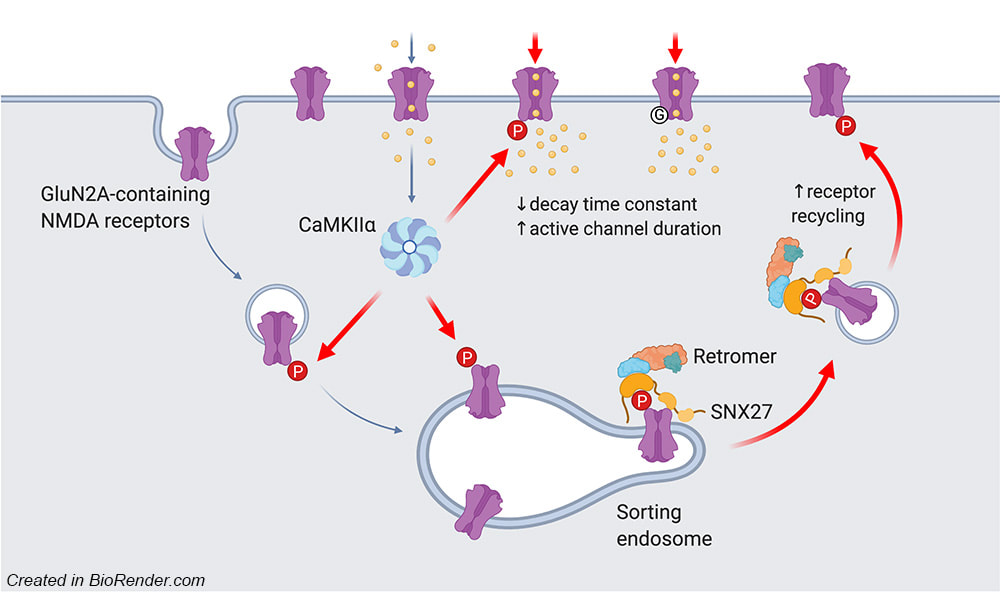
We are very excited to share our lab very first pre-print on bioRxiv. This work, led by our PhD student Hilary Yong, identified functional roles of CaMKIIα-mediated phosphorylation on the C-terminal tail of the GluN2A subunit (Ser-1459) in regulating the gating and activity-dependent trafficking of NMDA receptors. It is an extension of our recent collaborative work that was published in Cell Reports (Vieira et al., 2020). We thank all authors and our wonderful collaborators, Prof. Brett Collins (IMB, UQ), Dr. Angelo Keramidas (IMB, UQ), Prof. Joe Lynch (QBI, UQ) and Prof. Katherine Roche (NINDS, NIH, USA).
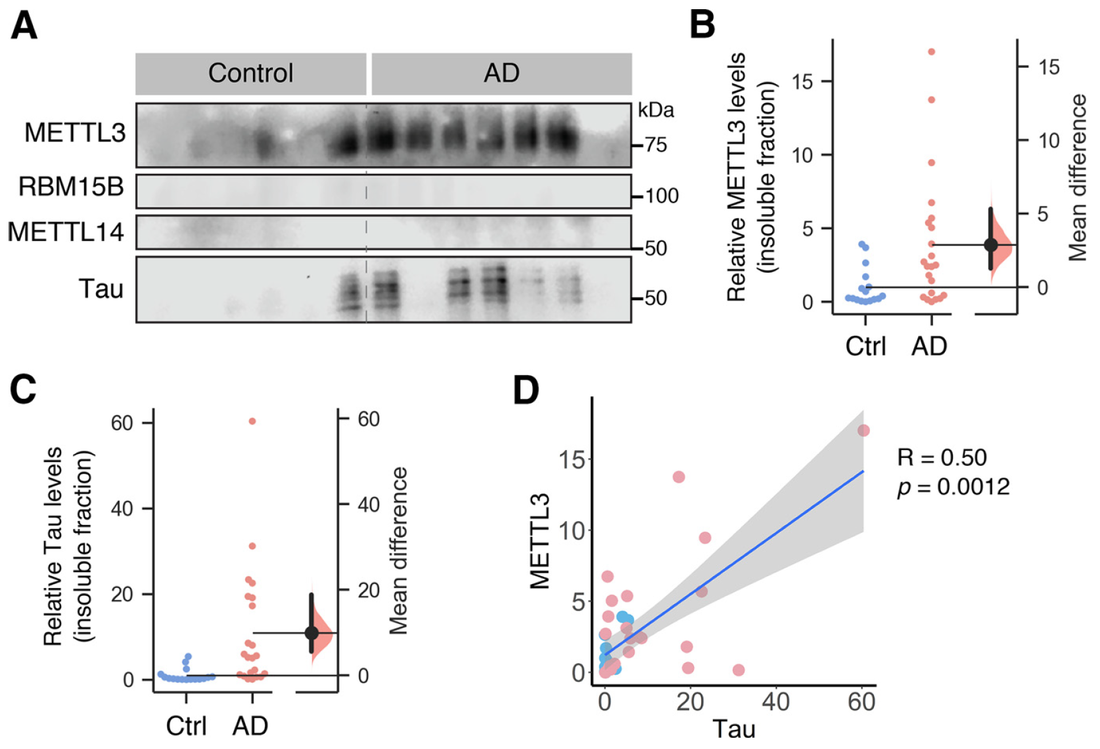
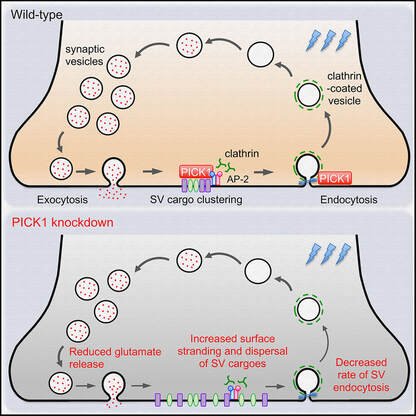 Our latest work has just been published in Cell Reports showing a new role for PICK1 (the only protein with a BAR and a PDZ domain) in regulating the activity-dependent clustering and retrieval of presynaptic vesicle cargo in mammalian central neurons. It is required for efficient synaptic vesicle endocytosis and sustained glutamate release. Outstanding work by Hilary and a wonderful collaboration with Prof. Mike Cousin (The University of Edinburgh, UK). 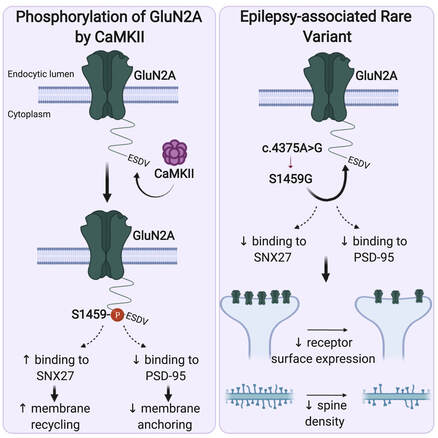 A new collaborative study led by the Roche's Lab at the National Institute of Neurological Disorders and Stroke (NINDS), National Institutes of Health (NIH), is now published in Cell Reports. This study identified CaMKIIα phosphorylation of the GluN2A subunit on Ser-1459 as a mechanism regulating NMDA receptor trafficking. An epilepsy-associated rare variant at this same residue, GluN2A-S1459G, results in altered protein interactions, decreased NMDA receptor surface expression, and reduced synaptic function, providing potential insight into an epilepsy phenotype. Congratulations to the lead author, Dr. Marta Vieira, and Katherine on this beautiful work. We are excited to share our new work, "Altered expression of the m6A methyltransferase METTL3 in Alzheimer's disease", which has just been published in eNeuro. Here we report a decrease in the expression of METTL3 mRNA and soluble protein in the postmortem hippocampal tissues of Alzheimer's disease (AD) patients. We also identified a striking alteration in the METLL3 protein expression, including enhanced insolubility and immunoreactivity in the AD hippocampus. Our results suggest that perturbation of m6A signalling may present a novel cellular mechanisms underpinning dysregulation of gene expression associated with AD pathophysiology. Congratulations to River and Jocelyn who led this study and to our QBI collaborators, Drs. Judith Camats-Perna and Rodrigo Medeiros.
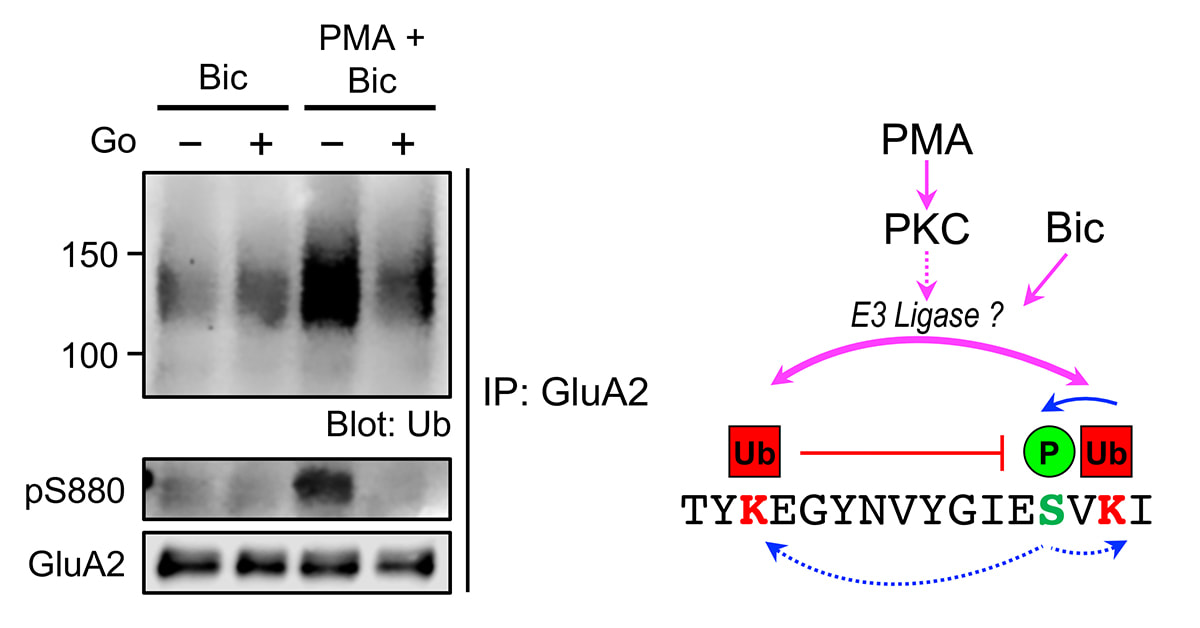
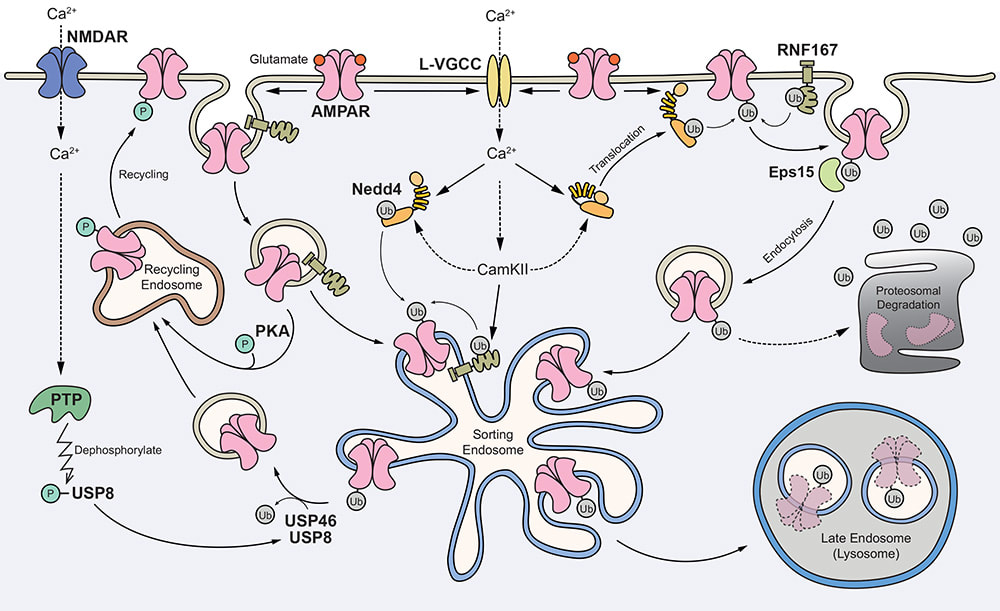
We are delighted that our study, primarily done by Jocelyn and Jun Wei, has just been published in Cellular and Molecular Neurobiology. Here, we demonstrated the cross-modulation between the ubiquitination and phosphorylation of the GluA2 subunit of AMPA receptors by protein kinase C (PKC). Interestingly, this effect is specific for GluA2 as phorbol ester does not potentiate bicuculline-induced ubiquitination of the GluA1 subunit. We envisage that the binding of glutamate on AMPARs in neurons with high level of PKC activity (such as following the activation of mGluRs) will result in subunit-specific regulation of AMPAR ubiquitination and intracellular sorting, which ultimately govern the subunit composition and number of AMPARs, including the Ca2+-permeable AMPARs at synapses.
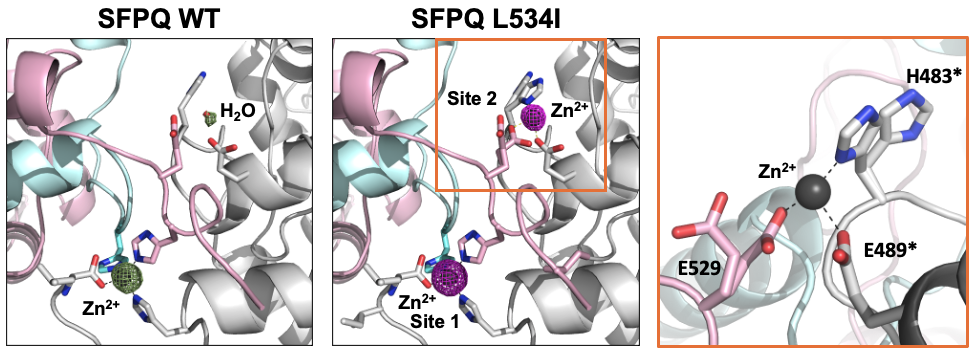
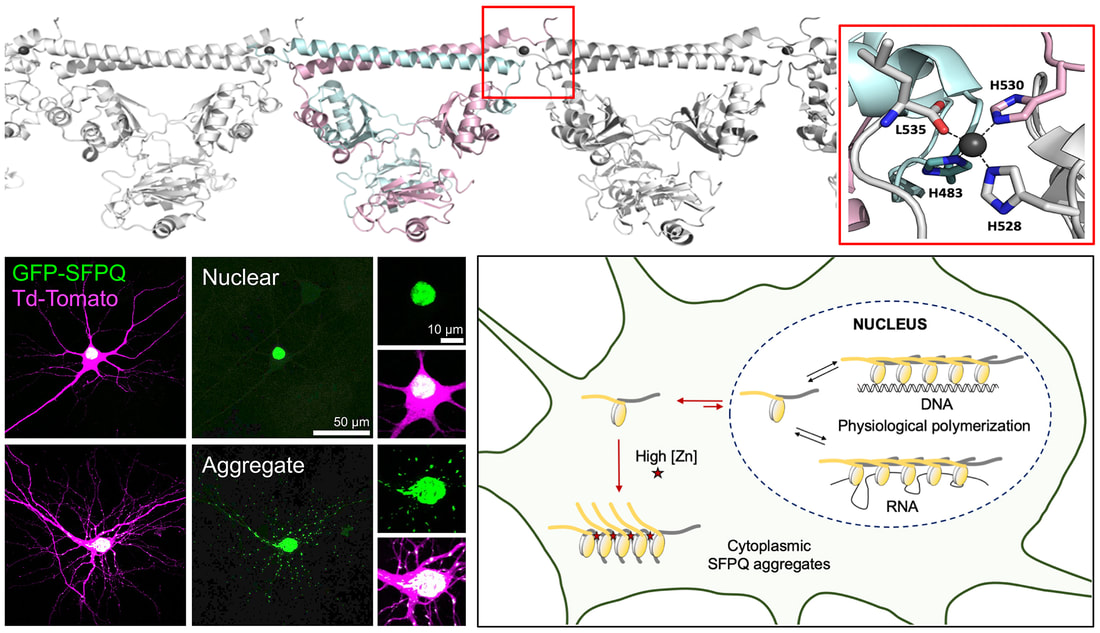
SFPQ is an abundant and ubiquitous nuclear RNA-binding protein (RBP) that has been implicated in gene regulation and subnuclear body formation. In a study led by Dr. Mihwa Lee (La Trobe University, Melbourne), we report the crystal structure of SFPQ in complex with Zn(II), which reveals an infinite polymer of SFPQ mediated by Zn(II) binding to the protein. The application of Zn(II) to primary cortical neurons induced the cytoplasmic accumulation and aggregation of SFPQ. Mutagenesis of the three Zn(II)-coordinating histidine residues resulted in a significant reduction in the zinc-binding affinity of SFPQ in solution and the Zn(II)-induced cytoplasmic aggregation of SFPQ in cultured neurons. This study, which was published in Nucleic Acids Research, offers a new framework for how metal-induced polymerization of RBPs can induce cytoplasmic aggregation, which are commonly associated with neurodegenerative diseases.
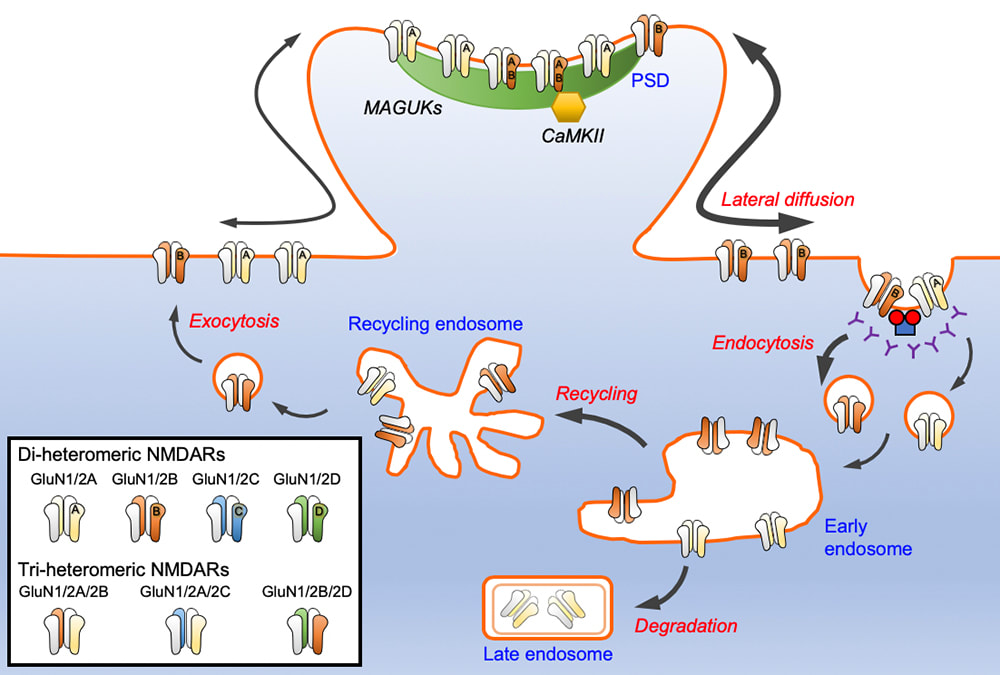
The N-methyl-D-aspartate glutamate receptors (NMDARs) mediate calcium-dependent signaling that underpins multiple forms of synaptic plasticity. Different GluN2 (GluN2A-D) subunit confers NMDARs with distinct ion channel properties and intracellular trafficking pathways. In a review article which has just been published in Journal of Neurochemistry, we discuss the current knowledge of the molecular mechanisms that regulate the trafficking of GluN2-containing NMDARs, focusing on the roles of several key synaptic proteins that interact via their carboxyl termini. This review article is a joint effort with our collaborator Prof. Katherine Roche (NINDS, NIH). Congratulations to Marta (Roche Lab) and Hilary (Anggono Lab) on an excellent work.
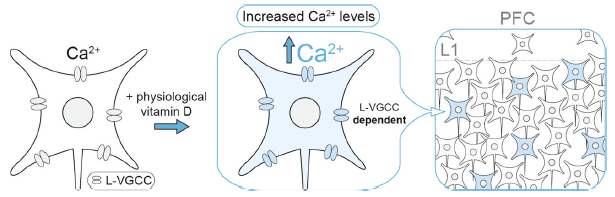
Genetic variants in genes encoding L-type voltage-gated calcium channel (L-VGCC) subtypes are associated with increased risk for schizophrenia. Likewise, epidemiological study has implicated developmental vitamin D deficiency as a risk factor for schizophrenia. In the latest study published in Translational Psychiatry, we (led by Prof. John McGrath of QBI, UQ and Aarhus University, Denmark) showed that the active vitamin D metabolite exert a rapid, non-genomic modulation of L-VGCCs in a subset of neurons in developing medial prefrontal cortex in mice. Optimal modulation of L-VGCCs by 1,25(OH)2-vitamin D may therefore contribute to the healthy development of Vitamin D-responsive neurons within the maturing cortical circuits.
A collaborative study led by a PhD student, Eunice Wong from the Degnan's Labs (School of Biological Sciences, UQ), has revealed ancient submodules of co-expressed "synaptic gene" orthologues during development and in cell type-specific manners. Although synapses do not exist in the sponge, these submodules may contribute to sensory roles in specific cell types in the sponge. The paper was published in the journal Scientific Reports.
Syntaxin1A neomorphic mutations promote rapid recovery from isoflurane anesthesia in fruit flies22/7/2019
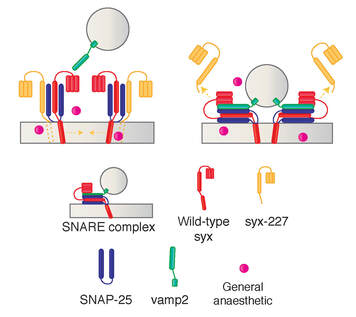 A collaborative study from the van Swinderen laboratory (QBI, UQ) published in Anesthesiology, has shown that transiently expressing a truncated syntaxin1A (Δ227) in adult Drosophila flies facilitates recovery from isoflurane anesthesia. Interestingly, our biochemical study revealed that the truncated syntaxin1A is absence from the presynaptic SNARE complex, suggesting that the resistance-promoting effect occurs prior to SNARE formation. We are proud to be part of a new study, published in Scientific Reports, confirming the link between neonatal vitamin D deficiency and the risk of schizophrenia. This study led by our collaborator Prof. John McGrath (QBI, UQ and Aarhus University, Denmark) was based on 2602 individuals (born between 1981-2000) from the Danish national registry. This is the largest study of its kind to date. [Press Release] [Infographic]
Transcriptional control of neuroblast migration in adult mouse ventricular-subventricular zone3/10/2018
A new collaborative paper in Cerebral Cortex led by the lab of A/Prof. Michael Piper (School of Biomedical Sciences, UQ) has revealed a role of the transcription factor NFIX (Nucleor Factor I X) in neuroblast migration within the adult mouse ventricular - subventricular zone. Conditional ablation of NFIX from neural stem or progenitor cells, or neuroblasts causes migration defects due to aberrant neuroblast branching, partly resulted from an increased expression of the guanylyl cyclase natriuretic peptide receptor 2 (Npr2). Congratulations to the team, especially Oressia and Lachlan from the Piper Lab!
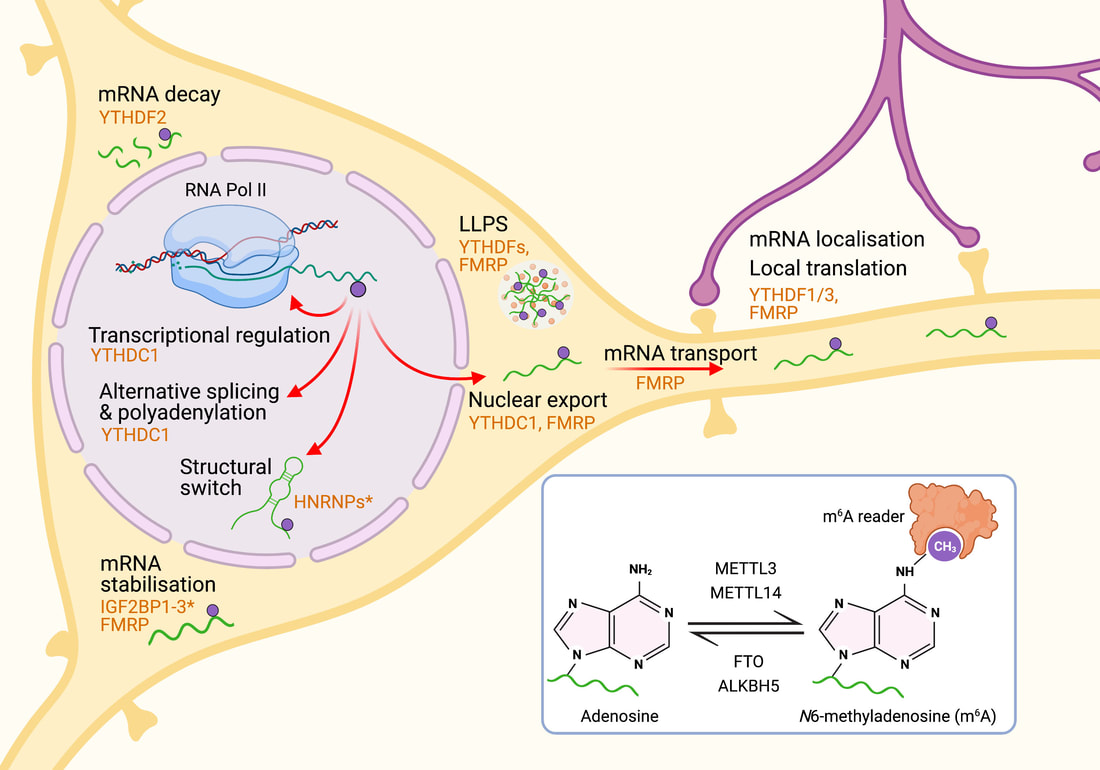
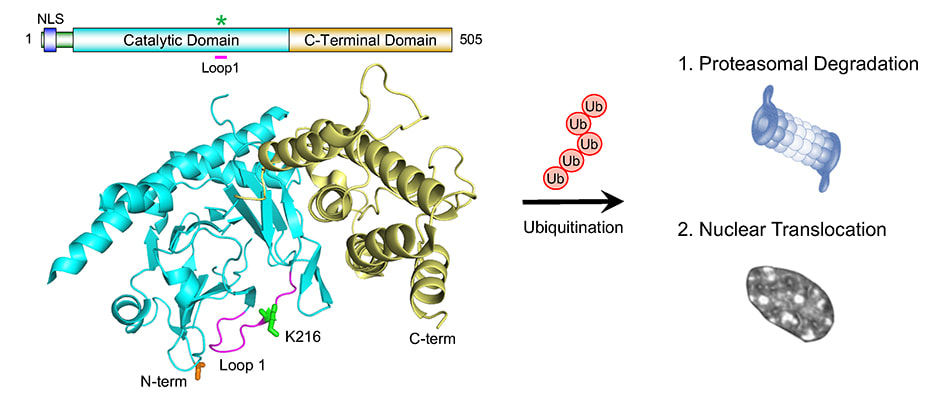
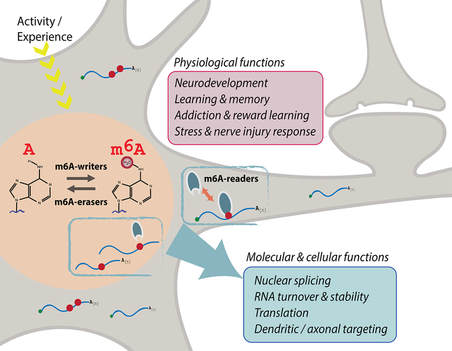 Jocelyn's new review article in the Journal of Neurochemistry is now online. N6-methyladenosine (m6A), the most prevalent internal modification on eukaryotic RNA, modulates transcript stability, splicing and translation efficiency. Activity-dependent m6A in the central nervous system provides a versatile and important mechanism to control RNA structure and function. This review article provides the current knowledge of m6A function and mechanism in neurobiology, highlighting the critical roles of m6A-epitranscriptomic regulation in multiple aspects of the mammalian nervous system, from neural development to plasticity in learning and memory.  Tianyi Zhu et al. (Volume 430/3) show that active ubiquitination of FTO mediates its proteasomal degradation, as well as its translocation into the nucleus during nutrient deprivation. The cover depicts an artistic rendition of FTO translocation into nuclei, which are stained by DAPI. The small puncta represent ubiquitinated FTO as revealed by the proximity ligation assay using anti-FTO and anti-ubiquitin antibodies. Cell artwork by Jocelyn Widagdo. Background visual design by Miriam Bucheli (Elsevier/Scientific Editor and Visual Designer). 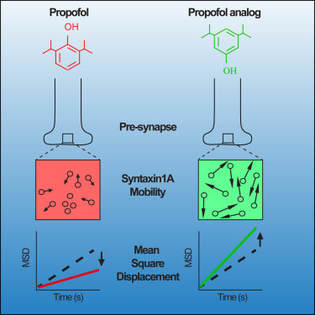 A major collaborative paper led by A/Prof. Bruno van Swinderen (QBI, UQ) has just been published in Cell Reports, describing the effect of a widely used general anaesthetic, propofol, in impairing the presynaptic release of neurotransmitters. Propofol restricts syntaxin 1a mobility on presynaptic membranes, a process that is dependent on another SNARE molecule, SNAP-25. Members of the Anggono Lab who were involved in this study include James Steeves (former Research Assistant) and Shu Liu (former Wen Zhou Scholar). [Press Release] Tianyi, Hilary and Jocelyn have just published a research article in the Journal of Molecular Biology, reporting that the fat mass and obesity-associated (FTO) protein undergoes post-translational ubiquitination at Lys-216 of the N-terminal catalytic domain. By using the CRISPR/Cas-9 approach, they further showed that FTO K216R knock-in cells have a slower rate of FTO protein turnover. In addition, these cells display a defect in FTO translocation into the nucleus following amino acid starvation. Great effort by the team and congrats!
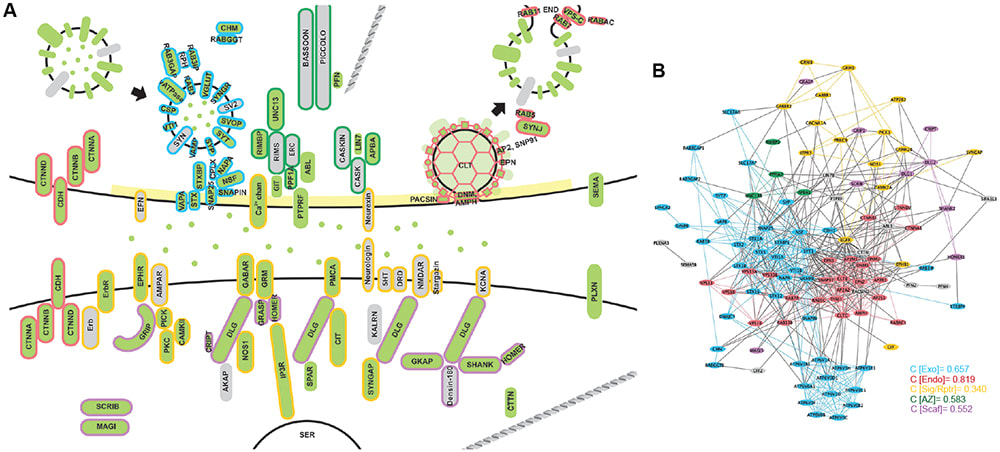
In this article, which has just been published in the Frontiers of Molecular Neuroscience, we review recent advances in the field and provide insights into the role of protein ubiquitination in regulating AMPA receptor trafficking and function in health and disease.
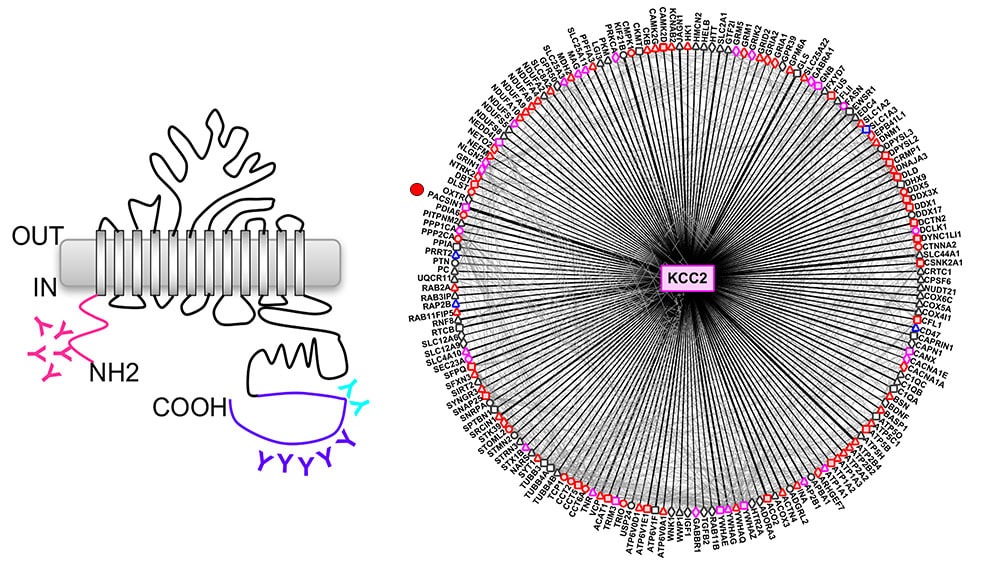
A major work from the Woodin Lab (University of Toronto) has just been published in eLife, in which Victor is a co-author. This is the first report describing the native KCC2 interactome in the brain. They found that KCC2 interacts with PACSIN1 (also known as syndapin1) to regulate synaptic inhibition in neurons.
Activation of GIRK channels by p75 neurotrophin receptors underlies Aβ-induced neuronal death26/7/2017
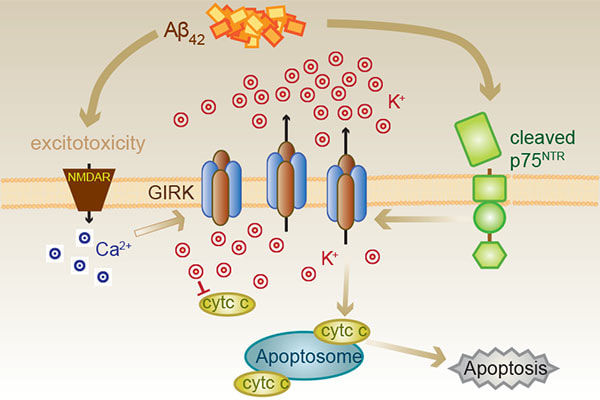
Joanne and Victor publish a collaborative paper with the Coulson Lab (UQ) in Frontiers in Neuroscience, reporting the role of G-protein-coupled Inwardly Rectifying Potassium (GIRK/Kir3) channels and p75 neurotrophin receptors in mediating potassium efflux during Aβ-induced neuronal death.
|
Synaptic Neurobiology LabArchives
January 2023
Categories
All
|
 RSS Feed
RSS Feed